|



[~][`] |
Lyell's "The Student's Elements of Geology"
Chapter 4
Introduction:
The Student's Elements of Geology
Chapter 1: On the Different Classes of Rocks
Chapter
2:
Aqueous Rocks
Chapter 3:
Fossils in Strata
Chapter 4:
Consolidation of Strata and Petrifaction
Chapter 5:
Strata Above the Sea
Chapter 6:
Denudation
CONSOLIDATION OF STRATA AND
PETRIFACTION OF FOSSILS
Chemical and Mechanical
Deposits. — Cementing together of Particles. — Hardening by Exposure to Air. —
Concretionary Nodules. — Consolidating Effects of Pressure. — Mineralization of
Organic Remains. — Impressions and Casts: how formed. — Fossil Wood. — Goppert’s
Experiments. — Precipitation of Stony Matter
most rapid where Putrefaction is going on. — Sources of Lime and Silex in
Solution.
Having spoken in
the preceding chapters of the characters of sedimentary formations, both as
dependent on the deposition of inorganic matter and the distribution of fossils,
I may next treat of the consolidation of stratified rocks, and the petrifaction
of imbedded organic remains.
Chemical and Mechanical Deposits.— A
distinction has been made by geologists between deposits of a mechanical, and
those of a chemical, origin. By the name mechanical are designated beds of mud,
sand, or pebbles produced by the action of running water, also accumulations of
stones and scoriæ thrown out by a volcano, which have fallen into their present
place by the force of gravitation. But the matter which forms a chemical deposit
has not been mechanically suspended in water, but in a state of solution until
separated by chemical action. In this manner carbonate of lime is occasionally
precipitated upon the bottom of lakes in a solid form, as may be well seen in
many parts of Italy, where mineral springs abound, and where the calcareous
stone, called travertin, is deposited. In these springs the lime is usually held
in solution by an excess of carbonic acid, or by heat if it be a hot spring,
until the water, on issuing from the earth, cools or loses part of its acid. The
calcareous matter then falls down in a solid state, incrusting shells, fragments
of wood and leaves, and binding them together.
That similar travertin is formed at some points in
the bed of the sea where calcareous springs issue can not be doubted, but as a
general rule the quantity of lime, according to Bischoff, spread through the
waters of the ocean is very small, the free carbonic acid gas in the same waters
being five times as much as is necessary to keep the lime in a fluid state.
Carbonate of lime, therefore, can rarely be precipitated at the bottom of the
sea by chemical action alone, but must be produced by vital agency as in the
case of coral reefs.
[This
reasoning - there are so few calcareous springs that large-scale rapid
deposition is not possible - would not be valid if the fountains of the deep
were broken up. In that instance vast carbonate deposits could be produced very
quickly. PRS]
In such reefs, large masses of limestone are formed
by the stony skeletons of zoophytes; and these, together with shells, become
cemented together by carbonate of lime, part of which is probably furnished to
the sea-water by the decomposition of dead corals. Even shells, of which the
animals are still living on these reefs, are very commonly found to be incrusted
over with a hard coating of limestone.
If sand and pebbles are carried by a river into the
sea, and these are bound together immediately by carbonate of lime, the deposit
may be described as of a mixed origin, partly chemical, and partly mechanical.
Now, the remarks already made in Chapter II, on the
original horizontality of strata are strictly applicable to mechanical deposits,
and only partially to those of a mixed nature. Such as are purely chemical may
be formed on a very steep slope, or may even incrust the vertical walls of a
fissure, and be of equal thickness throughout; but such deposits are of small
extent, and for the most part confined to vein-stones.
Consolidation of Strata.—It is chiefly in the
case of calcareous rocks that solidification takes place at the time of
deposition. But there are many deposits in which a cementing process comes into
operation long afterwards. We may sometimes observe, where the water of
ferruginous or calcareous springs has flowed through a bed of sand or gravel,
that iron or carbonate of lime has been deposited in the interstices between the
grains or pebbles, so that in certain places the whole has been bound together
into a stone, the same set of strata remaining in other parts loose and
incoherent.
Proofs of a similar cementing action are seen in a
rock at Kelloway, in Wiltshire. A peculiar band of sandy strata belonging to the
group called Oolite by geologists may be traced through several counties, the
sand being for the most part loose and unconsolidated, but becoming stony near
Kelloway. In this district there are numerous fossil shells which have
decomposed, having for the most part left only their casts. The calcareous
matter hence derived has evidently served, at some former period, as a cement to
the siliceous grains of sand, and thus a solid sandstone has been produced. If
we take fragments of many other argillaceous grits, retaining the casts of
shells, and plunge them into dilute muriatic or other acid, we see them
immediately changed into common sand and mud; the cement of lime, derived from
the shells, having been dissolved by the acid.
Traces of impressions and casts are often extremely
faint. In some loose sands of recent date we meet with shells in so advanced a
stage of decomposition as to crumble into powder when touched. It is clear that
water percolating such strata may soon remove the calcareous matter of the
shell; and unless circumstances cause the carbonate of lime to be again
deposited, the grains of sand will not be cemented together; in which case no
memorial of the fossil will remain.
In what manner silex and carbonate of lime may
become widely diffused in small quantities through the waters which permeate the
earth’s crust will be spoken of presently, when the petrifaction of fossil
bodies is considered; but I may remark here that such waters are always passing
in the case of thermal springs from hotter to colder parts of the interior of
the earth; and, as often as the temperature of the solvent is lowered, mineral
matter has a tendency to separate from it and solidify. Thus a stony cement is
often supplied to sand, pebbles, or any fragmentary mixture. In some
conglomerates, like the pudding-stone of Hertfordshire (a Lower Eocene deposit),
pebbles of flint and grains of sand are united by a siliceous cement so firmly,
that if a block be fractured, the rent passes as readily through the pebbles as
through the cement.
It is probable that many strata became solid at the
time when they emerged from the waters in which they were deposited, and when
they first formed a part of the dry land. A well-known fact seems to confirm
this idea: by far the greater number of the stones used for building and
road-making are much softer when first taken from the quarry than after they
have been long exposed to the air; and these, when once dried, may afterwards be
immersed for any length of time in water without becoming soft again. Hence it
is found desirable to shape the stones which are to be used in architecture
while they are yet soft and wet, and while they contain their “quarry-water,” as
it is called; also to break up stone intended for roads when soft, and then
leave it to dry in the air for months that it may harden. Such induration may
perhaps be accounted for by supposing the water, which penetrates the minutest
pores of rocks, to deposit, on evaporation, carbonate of lime, iron, silex, and
other minerals previously held in solution, and thereby to fill up the pores
partially. These particles, on crystallising, would not only be themselves
deprived of freedom of motion, but would also bind together other portions of
the rock which before were loosely aggregated. On the same principle wet sand
and mud become as hard as stone when
frozen; because one ingredient of the mass, namely,
the water, has crystallised, so as to hold firmly together all the separate
particles of which the loose mud and sand were composed.
Dr. MacCulloch mentions a sandstone in Skye, which
may be moulded like dough when first found; and some simple minerals, which are
rigid and as hard as glass in our cabinets, are often flexible and soft in their
native beds: this is the case with asbestos, sahlite, tremolite, and chalcedony,
and it is reported also to happen in the case of the beryl.*
The marl recently deposited at the bottom of Lake
Superior, in North America, is soft, and often filled with fresh-water shells;
but if a piece be taken up and dried, it becomes so hard that it can only be
broken by a smart blow of the hammer. If the lake, therefore, was drained, such
a deposit would be found to consist of strata of marlstone, like that observed
in many ancient European formations, and, like them, containing fresh-water
shells.
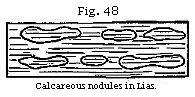
Concretionary Structure.—It is probable that
some of the heterogeneous materials which rivers transport to the sea may at
once set under water, like the artificial mixture called pozzolana, which
consists of fine volcanic sand charged with about twenty per cent of oxide of
iron, and the addition of a small quantity of lime. This substance hardens, and
becomes a solid stone in water, and was used by the Romans in constructing the
foundations of buildings in the sea. Consolidation in such cases is brought
about by the action of chemical affinity on finely comminuted matter previously
suspended in water. After deposition similar particles seem often to exert a
mutual attraction on each other, and congregate together in particular spots,
forming lumps, nodules, and concretions. Thus in many argillaceous deposits
there are calcareous balls, or spherical concretions, ranged in layers parallel
to the general stratification; an arrangement which took place after the shale
or marl had been thrown down in successive laminæ; for these laminæ are often
traceable through the concretions, remaining parallel to those of the
surrounding unconsolidated rock. (See Fig. 48.) Such nodules of limestone have
often a shell or other foreign body in the centre.
Among the most remarkable examples of concretionary
structure are those described by Professor Sedgwick as abounding in the
magnesian limestone of the north of England. The spherical balls are of various
sizes, from that of a pea to a diameter of several feet, and they have both a
concentric and radiated structure, while at the same time the laminæ of original
deposition pass uninterruptedly through them. In some cliffs this limestone
resembles a great irregular pile of cannon-balls. Some of the globular masses
have their centre in one stratum, while a portion of their exterior passes
through to the stratum above or below. Thus the larger spheroid in the section
(Fig. 49) passes from the stratum b upward into a. In this
instance we must suppose the deposition of a series of minor layers, first
forming the stratum b, and afterwards the incumbent stratum a;
then a movement of the particles took place, and the carbonates of lime and
magnesia separated from the more impure and mixed matter forming the still
unconsolidated parts of the stratum. Crystallisation, beginning at the centre,
must have gone on forming concentric coats around the original nucleus without
interfering with the laminated structure of the rock.
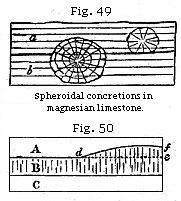
When the particles of rocks have been thus
rearranged by chemical forces, it is sometimes difficult or impossible to
ascertain whether certain lines of division are due to original deposition or to
the subsequent aggregation of several particles. Thus suppose three strata of
grit, A, B, C, are charged unequally with calcareous matter, and that B is the
most calcareous. If consolidation takes place in B, the concretionary action may
spread upward into a part of A, where the carbonate of lime is more abundant
than in the rest; so that a mass, d e f, forming a portion of the
superior stratum, becomes united with B into one solid mass of stone. The
original line of division, d e, being thus effaced, the line d f
would generally be considered as the surface of the bed B, though not strictly a
true plane of stratification.
Pressure and Heat.—When sand and mud sink to
the bottom of a deep sea, the particles are not pressed down by the enormous
weight of the incumbent ocean; for the water, which becomes mingled with the
sand and mud, resists pressure with a force equal to that of the column of fluid
above. The same happens in regard to organic remains which are filled with water
under great pressure as they sink, otherwise they would be immediately crushed
to pieces and flattened. Nevertheless, if the materials of a stratum remain in a
yielding state, and do not set or solidify, they will be gradually squeezed down
by the weight of other materials successively heaped upon them, just as soft
clay or loose sand on which a house is built may give way. By such downward
pressure particles of clay, sand, and marl may become packed into a smaller
space, and be made to cohere together permanently.
Analogous effects of condensation may arise when the
solid parts of the earth’s crust are forced in various directions by those
mechanical movements hereafter to be described, by which strata have been bent,
broken, and raised above the level of the sea. Rocks of more yielding materials
must often have been forced against others previously consolidated, and may thus
by compression have acquired a new structure. A recent discovery may help us to
comprehend how fine sediment derived from the detritus of rocks may be
solidified by mere pressure. The graphite or "black lead” of commerce having
become very scarce, Mr. Brockedon contrived a method by which the dust of the
purer portions of the mineral found in Borrowdale might be recomposed into a
mass as dense and compact as native graphite. The powder of graphite is first
carefully prepared and freed from air, and placed under a powerful press on a
strong steel die, with air-tight fittings. It is then struck several blows, each
of a power of 1000 tons; after which operation the powder is so perfectly
solidified that it can be cut for pencils, and exhibits when broken the same
texture as native graphite.
But the action of heat at various depths in the
earth is probably the most powerful of all causes in hardening sedimentary
strata. To this subject I shall refer again when treating of the metamorphic
rocks, and of the slaty and jointed structure.
Mineralisation of Organic Remains.—The
changes which fossil organic bodies have undergone since they were first
imbedded in rocks, throw much light on the consolidation of strata. Fossil
shells in some modern deposits have been scarcely altered in the course of
centuries, having simply lost a part of their animal matter. But in other cases
the shell has disappeared, and left an impression only of its exterior, or,
secondly, a cast of its interior form, or, thirdly, a cast of the shell itself,
the original matter of which has been removed. These different forms of
fossilisation may easily be understood if we examine the mud recently thrown out
from a pond or canal in which there are shells. If the mud be argillaceous, it
acquires consistency on drying, and on breaking open a portion of it we find
that each shell has left impressions of its external form. If we then remove the
shell itself, we find within a solid nucleus of clay, having the form of the
interior of the shell. This form is often very different from that of the outer
shell. Thus a cast such as a, Fig. 51, commonly called a fossil screw,
would never be suspected by an inexperienced conchologist to be the internal
shape of the fossil univalve, b, Fig. 51.
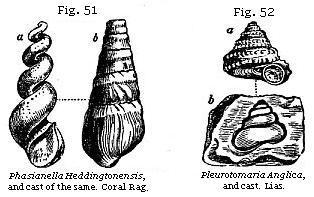
Nor should we have imagined at first sight that the
shell a and the cast b, Fig. 52, belong to one and the same fossil. The
reader will observe, in the last-mentioned figure (b, Fig. 52), that an
empty space shaded dark, which the shell itself once occupied, now
intervenes between the enveloping stone and the cast of the smooth interior of
the whorls. In such cases the shell has been dissolved and the component
particles removed by water percolating the rock. If the nucleus were taken out,
a hollow mould would remain, on which the external form of the shell with its
tubercles and striæ, as seen in a, Fig. 52, would be seen embossed. Now
if the space alluded to between the nucleus and the impression, instead of being
left empty, has been filled up with calcareous spar, flint, pyrites, or other
mineral, we then obtain from the mould an exact cast both of the external and
internal form of the original shell. In this manner silicified casts of shells
have been formed; and if the mud or sand of the nucleus happen to be incoherent,
or soluble in acid, we can then procure in flint an empty shell, which in shape
is the exact counterpart of the original. This cast may be compared to a bronze
statue, representing merely the superficial form, and not the internal
organisation; but there is another description of petrifaction by no means
uncommon, and of a much more wonderful kind, which may be compared to certain
anatomical models in wax, where not only the outward forms and features, but the
nerves, blood-vessels, and other internal organs are also shown. Thus we find
corals, originally calcareous, in which not only the general shape, but also the
minute and complicated internal organisation is retained in flint.

Such a process of petrifaction is still more
remarkably exhibited in fossil wood, in which we often perceive not only the
rings of annual growth, but all the minute vessels and medullary rays. Many of
the minute cells and fibres of plants, and even those spiral vessels which in
the living vegetable can only be discovered by the microscope, are preserved.
Among many instances, I may mention a fossil tree, seventy-two feet in length,
found at Gosforth, near Newcastle, in sandstone strata associated with coal. By
cutting a transverse slice so thin as to transmit light, and magnifying it about
fifty-five times, the texture, as seen in Fig. 53, is exhibited. A texture
equally minute and complicated has been observed in the wood of large trunks of
fossil trees found in the Craigleith quarry near Edinburgh, where the stone was
not in the slightest degree siliceous, but consisted chiefly of carbonate of
lime, with oxide of iron, alumina, and carbon. The parallel rows of vessels here
seen are the rings of annual growth, but in one part they are imperfectly
preserved, the wood having probably decayed before the mineralising matter had
penetrated to that portion of the tree.
[Such trees
are usually in a vertical position penetrating many horizontal strata. If
deposition really happened at eight centuries per inch the vast majority of the
tree would have decomposed and not been fossilized. PRS]
In attempting to explain the process of petrifaction
in such cases, we may first assume that strata are very generally permeated by
water charged with minute portions of calcareous, siliceous, and other earths in
solution. In what manner they become so impregnated will be afterwards
considered. If an organic substance is exposed in the open air to the action of
the sun and rain, it will in time putrefy, or be dissolved into its component
elements, consisting usually of oxygen, hydrogen, nitrogen, and carbon. These
will readily be absorbed by the atmosphere or be washed away by rain, so that
all vestiges of the dead animal or plant disappear. But if the same substances
be submerged in water, they decompose more gradually; and if buried in earth,
still more slowly; as in the familiar example of wooden piles or other buried
timber. Now, if as fast as each particle is set free by putrefaction in a fluid
or gaseous state, a particle equally minute of carbonate of lime, flint, or
other mineral, is at hand ready to be precipitated, we may imagine this
inorganic matter to take the place just before left unoccupied by the organic
molecule. In this manner a cast of the interior of certain vessels may first be
taken, and afterwards the more solid walls of the same may decay and suffer a
like transmutation. Yet when the whole is lapidified, it may not form one
homogeneous mass of stone or metal. Some of the original ligneous, osseous, or
other organic elements may remain mingled in certain parts, or the lapidifying
substance itself may be differently coloured at different times, or so
crystallised as to reflect light differently, and thus the texture of the
original body may be faithfully exhibited.
The student may perhaps ask whether, on chemical
principles, we have any ground to expect that mineral matter will be thrown down
precisely in those spots where organic decomposition is in progress? The
following curious experiments may serve to illustrate this point: Professor
Goppert of Breslau, with a view of imitating the natural process of
petrifaction, steeped a variety of animal and vegetable substances in waters,
some holding siliceous, others calcareous, others metallic matter in solution.
He found that in the period of a few weeks, or sometimes even days, the organic
bodies thus immersed were mineralised to a certain extent. Thus, for example,
thin vertical slices of deal, taken from the Scotch fir (Pinus sylvestris),
were immersed in a moderately strong solution of sulphate of iron. When they had
been thoroughly soaked in the liquid for several days they were dried and
exposed to a red-heat until the vegetable matter was burnt up and nothing
remained but an oxide of iron, which was found to have taken the form of the
deal so exactly that casts even of the dotted vessels peculiar to this family of
plants were distinctly visible under the microscope.
[So we see
fossilization in "several days" - long ages are not needed. PRS]
The late Dr. Turner observes, that when mineral
matter is in a “ nascent state,” that is to say, just liberated from a previous
state of chemical combination, it is most ready to unite with other matter, and
form a new chemical compound. Probably the particles or atoms just set free are
of extreme minuteness, and therefore move more freely, and are more ready to
obey any impulse of chemical affinity. Whatever be the cause, it clearly
follows, as before stated, that where organic matter newly imbedded in sediment
is decomposing, there will chemical changes take place most actively.
An analysis was lately made of the water which was
flowing off from the rich mud deposited by the Hooghly River in the Delta of the
Ganges after the annual inundation. This water was found to be highly charged
with carbonic acid holding lime in solution.**
Now if newly-deposited mud is thus proved to be permeated by mineral matter in a
state of solution, it is not difficult to perceive that decomposing organic
bodies, naturally imbedded in sediment, may as readily become petrified as the
substances artificially immersed by Professor Goppert in various fluid mixtures.
It is well known that the waters of all springs are
more or less charged with earthy, alkaline, or metallic ingredients derived from
the rocks and mineral veins through which they percolate. Silex is especially
abundant in hot springs, and carbonate of lime is almost always present in
greater or less quantity. The materials for the petrifaction of organic remains
are, therefore, usually at hand in a state of chemical solution wherever organic
remains are imbedded in new strata.
[When the fountains of the deep were broken up we would therefore have ideal
conditions for large-scale fossilization. PRS]
*
Dr. MacCulloch, Syst. of Geol., vol. i, p. 123.
**
Piddington, Asiat. Research., vol. xviii, p. 226.
|
|